
Date: 24 August 2016 (Wed)
Time: 3:00 pm
Venue: INFORMM Auditorium, USM Main Campus
There will be a seminar by our visiting scientists from Japan, Dr. Li-Foong Yoong a Research Scientist at the Laboratory for Genetic Control of Neuronal Architecture at RIKEN Brain Science Institute and Assoc. Prof. Dr. Itsuki Ajioka from the Center for Brain Integration Research at Tokyo Medical and Dental University.
Summaries
1. How do neurons build their complex dendrite arbors?
Li-Foong Yoong, Ph.D.
Research Scientist
Laboratory for Genetic Control of Neuronal Architecture
RIKEN Brain Science Institute, JAPAN
Dendrites are the crucial structure of neurons where signals are received and propagated. Neurons require highly branched dendritic arbor morphologies to wire together neural networks and define the individual functional properties of the cell. The intricate cell biological mechanisms by which such complex dendritic arbor architectures are established remain largely unknown. Importantly these mechanisms are often disrupted in neurological disorders and may be utilized during neuronal regeneration. To understand how final morphology is achieved, assaying not just the final dendrite shape but rather the continuous imaging of neurons during dendrite outgrowth and arborization is required. However, live imaging of mammalian neuron has been proven challenging as they take weeks and sometime months to develop. On the other hand, insect develops and matures its nervous system in just days. Taking advantage of this system, our lab uses Drosophila melanogaster as a model for deciphering the molecular mechanisms underlying the complex dendrite arborization processes (Nature Neuroscience. 2015;18(10):1437-45, Nature Neuroscience. 2011;15(2):224-33, Neuron. 2007;56(6):963-78.)
In this talk, I will discuss our recent work of studying complex dendrite formation using a novel non-invasive, in vivo time-lapse microscopy approach. We performed live imaging-based RNAi screening coupled with computer vision-based quantification, and have identified groups of signaling molecules and cytoskeletal modulators that control the dynamics of the dendrite development processes.
2. Cell-cycle and Biomaterial Engineering for Injured Brain Regeneration
Itsuki Ajioka, Ph.D.
Center for Brain Integration Research
Tokyo Medical and Dental University, JAPAN
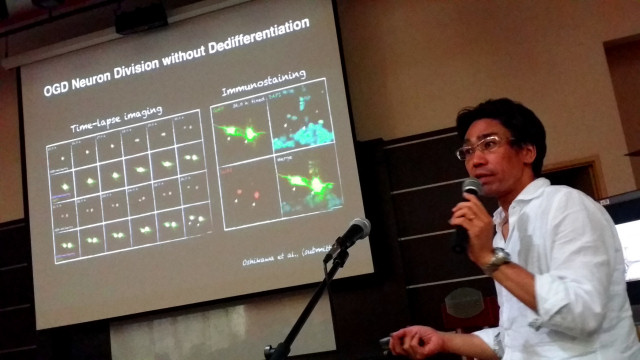
Our laboratory is focusing on injured brain regeneration from the view of biology (developmental neurobiology) and bioengineering (biomaterial engineering). Although our brain do not regenerate after injury by self-healing, recent advances in neurobiology uncovered the potency of brain regeneration. For example, we found and reported the proliferation potency of neurons (Ajioka et al., Cell 2007; Oshikawa et al., Development 2013). How can we bring out the regenerative potential in injured brain? We tried to understand the regeneration potency of injured brain by the approach of “science”, which reflects building and organizing knowledge of something that does exist. In contrast to “science”, “engineering” reflects a creative application for the development of something that does not exist. Thus, “engineering” approach could be important to bring out the regenerative potential in injured brain. For example, we developed artificial scaffolds and reported the technique to proliferate neurons in a 3D manner (Ajioka et al., Biomaterials 2011) and the technique to align neurons in the injured brain region (Ajioka et al., Tissue Eng Part A 2015). In this talk, I will introduce our recent finding on injured brain regeneration.
Update:
Dr. Yoong Li Foong

Dr. Itsuki Ajioka
URICAS members with the speakers.










 Prof. Dr. Toru Takumi, MD, PhD
Prof. Dr. Toru Takumi, MD, PhD


